
When heated to decomposition it emits toxic fumes of K 2O and SO x. Swallowing large doses causes severe gastrointestinal tract effects. Moderately toxic experimentally by subcutaneous route. S24/25: Avoid contact with skin and eyes. 
Safety Statements of Potassium sulfate?(CAS NO.): 22-24/25? 4.2 Caution Statement P264, P280, P305+P351+P338, P310, P33, P313 4.2 Formulations/Preparations GRADES: HIGHEST PURITY MEDICINAL COMMERCIAL CRUDE CP AGRICULTURALĬommercial potassium sulfate contains not less than 48% soluble potash (K2O) chiefly as sulfate, and not more than 2.5% chlorine 4.3 WGK Germany 1 4.3 RTECS TT5900000 4.3 Report When heated todecomposition it emits toxic fumes of K2Oand SOx. Moderately toxicexperimentally by subcutaneous route.Swallowing large doses causes severegastrointestinal tract effects. 3.3 Usage Technical grades are used in fertilizers for manufacture of potassium alum, potassium carbonate and glass the reagent grade is used in the Kjeldahl determination of nitrogen.Ĥ.1 Risk Statements S22-S24/25 4.1 Safety Statements S22 S24/25 4.1 Hazard Declaration H318 4.1 RIDADR NONH for all modes of transport 4.1 Safety Profile Moderately toxic tohumans by ingestion. 3.2 Purification Methods Potassium sulfate M 174.3, m 1069o, d 4 2.67 It crystallised from distilled water (4mL/g at 20o 8mL/g at 100o) between 100o and 0o. SULFATE HAS ALSO BEEN MADE FROM CEMENT MILL DUST, LANGBEINITE, & FROM MURIATE BY TREATMENT WITH SODIUM OR MAGNESIUM SULFATE OR WITH SULFURIC ACID. (B) BY FRACTIONAL CRYSTALLIZATION OF NATURAL SULFATE ORE (C) FROM SALT LAKE BRINES.|THE POTASSIUM SALT OF SULFURIC ACID.FIRST POTASH SALT PRODUCED COMMERCIALLY IN US FROM ANYTHING OTHER THAN WOOD ASHES. 1.10 Isomers Smiles S(=O)(=O).ģ.1 Methods of Manufacturing REACTION OF POTASSIUM CHLORIDE WITH LANGBEINITE ORE, SCHOENITE OBTAINED FROM KAINITE ORE, OR SODIUM SULFATE (GLASERITE IS AN ISOLATED INTERMEDIATE) REACTION OF POTASSIUM CHLORIDE WITH SULFURIC ACID OR SULFUR DIOXIDE, WATER AND OXYGEN RECOVERY FROM SUGAR WASTES|(A) BY TREATMENT OF POTASSIUM CHLORIDE EITHER WITH SULFURIC ACID OR WITH SULFUR DIOXIDE, AIR, & WATER (HARGREAVES PROCESS). Arcanum duplicatum arcanumduplicatum Dipotassium sulfate EINECS 231-915-5 Glazier's salt K2SO4 Kalii sulfas Kalium sulphuricum Kaliumsulfat kaliumsulphuricum KJELTABS IB 61 MFCD03456797 Monopotassium sulfate POTASSIUM SULFATE ANHY LOW NIT Potassium sulfate test solution(ChP) POTASSIUM SULFATE, BIO-REFINEDPOTASSIUM SULFATE, BIO-REFINEDPOTASSIUM SULFATE, BIO-REFINEDPOTASSIUM SULFATE, BIO-REFINED POTASSIUM SULFATE, GRANULAR, REAGENT (ACS)POTASSIUM SULFATE, GRANULAR, REAGENT (ACS)POTASSIUM SULFATE, GRANULAR, REAGENT (ACS) POTASSIUM SULFATE, POWDER, REAGENT (ACS)POTASSIUM SULFATE, POWDER, REAGENT (ACS)POTASSIUM SULFATE, POWDER, REAGENT (ACS)POTASSIUM SULFATE, POWDER, REAGENT (ACS) potassium sulphate salpolychrestum Salt of leMery sop Sulfuric acid dipotassium salt Sulfuric acid, dipotassium salt View all 1.3 CAS No. S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible). S36/37/39:Wear suitable protective clothing, gloves and eye/face protection. S28:After contact with skin, wash immediately with plenty of. S27:Take off immediately all contaminated clothing. S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. R20/21/22:Harmful by inhalation, in contact with skin and if swallowed. It is involved in the preparation of chrome catalyst. It is also used in preparation of chromium metal, dye, in printing and dyeing industry. 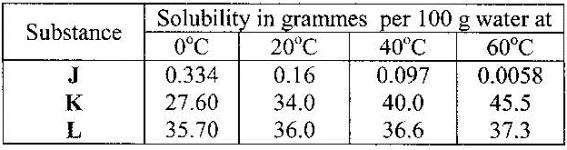
Greenish black powder(s) amorphous Ĭhromium(III) sulfate hydrate is widely used in making chrome tanned leather, paints, inks and ceramics. Chromium(iii) sulfate hydrate, puratronicĬhromium(iii) sulfate hydrate, reagent gradeĬhromium(III) sulfate hydrate, Puratronic(R), 99.999% (metals basis)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
